Hip pain is a common issue faced by adults in recent times. There can be several reasons for hip pain like over-exercising, any injury, or sprain. Mostly the pain settles after some days but if the pain occurs frequently and starts to get severe, it can affect a person’s quality of life and day-to-day behavior. It is crucial to visit a doctor and get medical advice as the hip pain may also be due to a bone disease called avascular necrosis (AVN), also known as osteonecrosis.
What is avascular necrosis (AVN)?
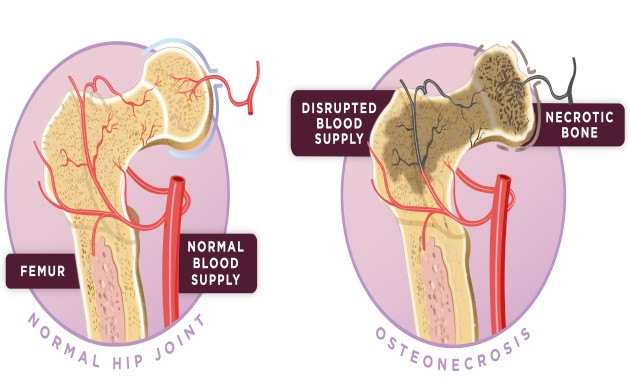
Bones in our body continuously rebuild themselves to stay healthy. In AVN, the bones lose their ability to rebuild due to the interruption of blood supply and hence the bone cells get damaged. Common day-to-day activities such as walking, standing, climbing stairs, lifting moderate to heavy objects, and full movement of affected joints become difficult. Pain may sometimes persist even while resting. Any bone can be affected by AVN but it mostly occurs in the hip joint.
People in the age range of 20 to 50 years are at higher risk of AVN. AVN is akin to cancer because, like cancer, AVN progresses in different stages. If AVN isn’t diagnosed and treated on time, it can result in limiting the range of motion of the joint and can cause sudden and complete joint failure resulting in permanent disability. In severe cases, patients must undergo total hip replacement which can have additional complications.
What are the symptoms of AVN?
In the early stages, there is minimal or no pain. As AVN progresses, it may hurt when pressure is put on the affected bones. Most common symptoms include:
Severe pain in the hip joint: Joint pain can range from mildly irritating to unbearable. The pain can progress to areas surrounding the hip as well.
Stiffness causing loss of function of the joint: Activities like bending, rotation, and squatting become excruciatingly painful.
Limping: It can be difficult to walk normally with mild to severe AVN. The joint cannot bear the body’s weight which leads to difficulty in walking.
What causes AVN?
The most common causes of AVN are:
Alcohol abuse: Individuals drinking greater than 400mL of alcohol per week are at risk of AVN. Prolonged alcohol consumption results in an increase in the fatty acid concentration of the body leading to bone death.
Excess steroid intake: Use of steroids such as corticosteroids disturbs the body’s natural repair mechanism which damages the bones.
Accidents: Traumatic injuries causing fractures around the hip joint lead to increased stress in the bone that can lead to joint impairment.
Idiopathic: In many cases, a cause cannot be identified, and these patients receive the designation of idiopathic AVN.
How is AVN diagnosed?
Hip pain is the first sign of AVN. Usually, when AVN is suspected, an X-ray scan is recommended first. Once the diagnosis has been made, Magnetic Resonance Imaging (MRI) scan is used to determine the extent of tissue damage and the stage of AVN (Stage 1-Stage 4).
OSSGROW® – bone cell therapy for AVN
When cells in the body cannot self-repair, cell therapy can be useful for replacing them with newer and healthier cells. In AVN, bone-forming cells are damaged and lose their ability to rebuild themselves. During bone cell therapy, new bone cells are implanted by the doctor into the damaged area so that new bone is formed which restores the normal functions of the hip joint. The newly formed 3-dimensional bone will be useful in arresting the progression of the bone damage due to AVN and also enable free joint movement.
Regrow Biosciences® Pvt. Ltd. has developed a cell therapy product – OSSGROW® for the treatment of AVN that involves harvesting patients’ own healthy cells and implanting them back into the affected area. The new bone cells have the capacity to rebuild the bones and resume normal day-to-day activities. OSSGROW® is the only treatment option available for AVN.
Here’s everything you need to know about OSSGROW® – the bone cell treatment of AVN. Learn more here.
Benefits of OSSGROW® bone cell therapy:
Patient’s own cells are used, it’s a natural treatment
– It is a one-time treatment
– Arrests disease progression
– Less invasive treatment
– Less dependence on medication
– Prevention of highly invasive total hip replacement surgery
– Patients can resume normal life after the treatment
– Lifetime therapeutic efficiency
Key facts about OSSGROW®
– Only approved treatment option available for AVN.
– One-time treatment with ZERO side effects.
– Totally safe, and clinically tested cell therapy product.
– Market authorization and manufacturing license by the Drug Controller General of India, Ministry of Health and Family Welfare, Govt. of India
– OSSGROW® is covered under cashless and reimbursement schemes for all major Private/ Government and State Health Insurance Schemes.
For more information, contact 1800-209-0309 or visit www.ossgrow.com or www.regrow.in
Disclaimer: This is a sponsored article powered by Regrow Biosciences Pvt. Ltd. (“Regrow®”). Tata 1mg does not endorse any brands and is not responsible for their efficacy. This article powered by Regrow provides basic information on Avascular Necrosis (AVN) and Bone Cell Therapy (OSSGROW®) for the treatment of AVN. The intent of this article is to educate the general public at large on AVN and Bone Cell Therapy (OSSGROW®). Your perusal of this article does not establish a doctor-patient relationship. The information provided in this article is intended for general knowledge and is not an attempt to practice medicine or provide specific medical advice or a substitute for professional medical advice or treatment for medical conditions and it should not be used to make a diagnosis or to replace or overrule a qualified healthcare provider’s judgment.
Please consult your healthcare provider to diagnose any health problems and to answer any questions or concerns you may have regarding your condition. All efforts have been made to provide information in this article as accurate as possible. Although the content of this article is generally reviewed and Bone Cell Therapy (OSSGROW®) is approved by Indian FDA, Tata 1mg and Regrow do not make any representation or warranties or take any responsibility or liability with respect to the accuracy, applicability, timeliness, fitness or completeness of the contents of this article, and any other warranty, express or implied, or for any errors or omissions in this article, for the results obtained from its use, or for any damage or loss caused or alleged to be caused by reliance on the information available in this article.