
ग्लाइमेस्टर एम4 टैबलेट पीआर
लेखक डॉ. लिपिका खुराना, पीजीडीएचएचएम, बीडीएस,
समीक्षक डॉ. मेखला चंद्रा, MD, MBBS,
लास्ट अपडेटेड on 07 जन 2026 | 03:32 पीएम (Ist)

अतिरिक्त ऑफ़र के साथ अधिक बचत करें









सामान्य जानकारी
शामिल है
मार्केटर
स्टोरेज के निर्देश

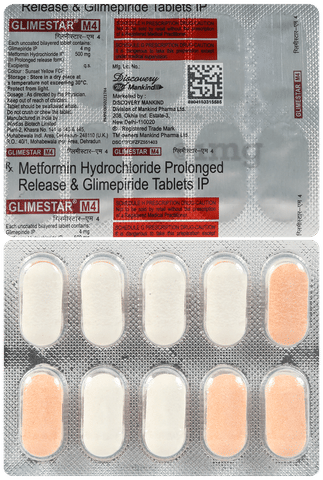
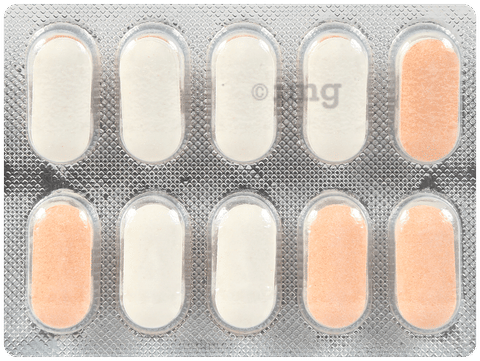


परिचय
ग्लाइमेस्टर एम4 टैबलेट पीआर को भोजन के साथ लेना चाहिए. अधिकतम लाभ प्राप्त करने के लिए इसे हर रोज एक नियमित रूप से एक ही समय पर लें. आपका डॉक्टर यह तय करेगा कि आपके लिए कौन सी खुराक सबसे अच्छी है, और यह आपके ब्लड शुगर लेवल के अनुसार, समय-समय पर कैसे काम कर रहा है, इसके अनुसार बदल सकता है.
अगर आप अच्छा महसूस कर रहे हैं या आपके ब्लड शुगर का स्तर नियंत्रित है, तो भी इस दवा का सेवन करते रहें. अगर आप इसे डॉक्टर से परामर्श किए बिना लेना बंद करते हैं, तो आपके ब्लड शुगर का स्तर बढ़ सकता है और आपको किडनी का नुकसान, अंधापन, नसों से संबंधित समस्याएं और अंगों को खोने का जोखिम रहता है. याद रखें कि यह दवा, इलाज का केवल एक हिस्सा है जिसमें आपके डॉक्टर द्वारा दी गई सलाह के अनुसार स्वस्थ आहार और नियमित व्यायाम भी शामिल होना चाहिए. आपकी जीवनशैली डायबिटीज को नियंत्रित करने में एक बड़ी भूमिका निभाती है.
ग्लाइमेस्टर एम4 टैबलेट पीआर का सबसे सामान्य साइड इफेक्ट ब्लड ग्लूकोज स्तर में गिरावट (हाइपोग्लाइसेमिया) है. ब्लड ग्लूकोज़ लेवल कम होने के लक्षणों को पहचानना ज़रूरी है, जैसे पसीना आना, चक्कर आना, सिरदर्द और हाथों का कांपना, और यह भी जानना ज़रूरी है कि ऐसी स्थिति में क्या करना चाहिए. इससे बचने के लिए, नियमित भोजन करना और हमेशा ग्लूकोज का तेज़ी से काम करने वाला स्रोत, जैसे शुगर फूड या फ्रूट जूस, आपके साथ रखना महत्वपूर्ण है. शराब पीने से आपका ब्लड शुगर के स्तर में कमी का जोखिम भी बढ़ सकता है और इसलिए इससे बचना चाहिए. इस दवा को लेने पर जो अन्य दुष्प्रभाव देखे जा सकते हैं उनमें स्वाद में बदलाव, मिचली आना, डायरिया, पेट दर्द, सिरदर्द और ऊपरी श्वसन पथ में संक्रमण शामिल हैं. इस दवा से कुछ लोगों का वजन बढ़ सकता है.
अगर आपको टाइप 1 डायबिटीज, एक्यूट या क्रोनिक मेटाबोलिक एसिडोसिस हो, जिसमें डायबिटिक कीटोएसिडोसिस भी शामिल है, तो इसे न लें. इस दवा को लेने से पहले, अगर आपको कभी हृदय रोग था तो अपने डॉक्टर को बताएं. यह उपयुक्त नहीं हो सकता है. गर्भवती या स्तनपान कराने वाली महिलाओं को इसे लेने से पहले डॉक्टर से भी परामर्श करना चाहिए. आपका ब्लड शुगर लेवल नियमित रूप से चेक किया जाना चाहिए, और आपका डॉक्टर आपके ब्लड सेल काउंट और लिवर फंक्शन की निगरानी करने के लिए ब्लड टेस्ट की सलाह भी दे सकता है.
ग्लिमेस्टर-एम टैबलेट पीआर के मुख्य इस्तेमाल
ग्लिमेस्टर-एम टैबलेट पीआर के फायदे
ग्लाइमेस्टर एम4 टैबलेट पीआर एक कॉम्बिनेशन दवा है जो आपके शरीर के द्वारा बनाए जाने वाले इन्सुलिन की मात्रा को (अग्नाशय में) बढ़ाती है. इसके बाद इंसुलिन आपके ब्लड ग्लूकोज़ लेवल को कम करने के लिए काम करता है. आमतौर पर इसे दिन में एक बार लिया जाता है. निर्धारित अवधि तक इसका सेवन जारी रखें.
ब्लड ग्लूकोज के लेवल को कम करना डायबिटीज को नियंत्रित करने का प्रमुख हिस्सा है. अगर आप इन स्तरों को नियंत्रित कर सकते हैं, तो आपमें डायबिटीज के कारण होने वाली गंभीर जटिलताओं जैसे कि किडनी का नुकसान, आंखों में नुकसान, तंत्रिका संबंधी समस्याएं और हाथ-पैरों का नुकसान आदि जैसे जोखिमों की संभावनाएं कम हो जाएगी. उचित आहार और व्यायाम के साथ इस दवा का नियमित सेवन आपको स्वस्थ और सामान्य जीवन जीने में मदद करेगा.
ग्लिमेस्टर-एम टैबलेट पीआर के साइड इफेक्ट
- सिरदर्द
- हाइपोग्लाइसीमिया (लो ब्लड ग्लूकोज लेवल)
- मिचली आना
- डायरिया
ग्लिमेस्टर-एम टैबलेट पीआर का इस्तेमाल कैसे करें
ग्लिमेस्टर-एम टैबलेट पीआर किस प्रकार काम करता है
All substitutes for Glimestar M4 Tablet PR
GlUcOnorm-G 4 Tablet PR
Zoryl-M4 Tablet PR
Azulix 4 MF Tablet PR
Isryl-M 4 Tablet PR
Obimet GX 4 Tablet PR
ख़ास टिप्स
- आपको नियमित व्यायाम, पौष्टिक भोजन और डायबिटीज की अन्य दवाइयों के साथ ग्लाइमेस्टर एम4 टैबलेट पीआर का सेवन जारी रखना चाहिए.
- पेट खराब होने की संभावना कम करने के लिए इसे भोजन के साथ लें.
- जब आप इस दवा का सेवन कर रहे हों तो अपने ब्लड शुगर लेवल की नियमित जांच करें.
- यदि आप इसे अन्य एंटीडायबिटीज दवाओं, शराब के साथ इस्तेमाल करते हैं या अगर आप भोजन में देरी करते हैं या नहीं खाते हैं तो इसके कारण आपको हाइपोग्लाइसीमिया (लो ब्लड शुगर लेवल) हो सकता है.
- अगर आप कोई सर्जरी कराने जा रहे हैं जिसमें जनरल एनेस्थेटिक का प्रयोग होगा, तो डॉक्टर को अपने डायबिटीज के इलाज के बारे में बताएं.
- अगर आपको तेज या गहरी सांस लेने की समस्या हो, या मिचली आना, उल्टी और पेट दर्द लगातार बना हुआ हो, तो तुरंत अपने डॉक्टर को बताएं, क्योंकि ग्लाइमेस्टर एम4 टैबलेट पीआर एक दुर्लभ लेकिन गंभीर स्थिति लैक्टिक एसिडोसिस का कारण बन सकता है, जो रक्त में लैक्टिक एसिड की अधिकता है.
- आपका डॉक्टर नियमित रूप से आपका लिवर फंक्शन चेक कर सकता है. अगर आपको पेट दर्द, भूख न लगना, या आंख या त्वचा का पीला (पीलिया) होना जैसे लक्षण दिखें तो अपने डॉक्टर को बताएं.
सुरक्षा संबंधी सलाह

अल्कोहल

गर्भावस्था

स्तनपान

ड्राइविंग
किडनी
गंभीर किडनी की बीमारी से पीड़ित मरीजों को ग्लाइमेस्टर एम4 टैबलेट पीआर का इस्तेमाल करने की सलाह नहीं दी जाती है. इस दवा का सेवन करते समय किडनी फंक्शन टेस्ट की नियमित निगरानी की सलाह दी जाती है.

लिवर
ग्लाइमेस्टर एम4 टैबलेट पीआर को आमतौर पर हल्के से मध्यम लिवर की बीमारी वाले मरीजों में कम खुराक के साथ शुरू किया जाता है, और लिवर की गंभीर बीमारी से पीड़ित मरीजों के लिए इसका इस्तेमाल न करने की सलाह दी जाती है.
अक्सर पूछे जाने वाले प्रश्न
फैक्ट बॉक्स
लत लगने की संभावना
चिकित्सीय वर्ग
संदर्भ
- Glimepiride [Prescribing Information]. Bridgewater, New Jersey: Sanofi-Aventis U.S. LLC.; 2009. [Accessed 08 Apr. 2019] (online) Available from:
- Metformin hydrochloride [Prescribing Information]. Princeton, New Jersey: Bristol-Myers Squibb Company. [Accessed 08 Apr. 2019] (online) Available from:
- American Diabetes Association. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes-2020. Diabetes Care. 2020;43(Suppl 1): S98-S110. [Accessed 28 Oct. 2021] (online) Available from:




यूजर का फीडबैक
और अधिक जानना चाहते हैं
हम आपको प्रमाणिक, भरोसेमंद और प्रासंगिक जानकारी प्रदान करते हैंक्या आपको यहां दी गई जानकारी से कोई समस्या है?
डिस्क्लेमर
मार्केटर की जानकारी
Address: 208, ओखला इंडस्ट्रियल एस्टेट, फेज III, नई दिल्ली - 110020
विक्रेता विवरण
A लाइसेंस वेंडर पार्टनरआपकी सबसे नज़दीकी लोकेशन से ग्लाइमेस्टर एम4 टैबलेट पीआर डिलीवर करेगा. जैसे ही फार्मेसी आपका ऑर्डर स्वीकार कर लेती है, फार्मेसी का विवरण आपके साथ शेयर किया जाएगा. आपके ऑर्डर की स्वीकृति आपके डॉक्टर की ℞ की वैधता और इस दवा की उपलब्धता पर आधारित है.
किसी भी समस्या के मामले में, हमसे संपर्क करें
ईमेल आईडी: [email protected]फोन नंबर: 0124-4166666
पता: 5th फ्लोर, प्रेसीडेंसी बिल्डिंग टावर-बी, 46/4 महरौली गुडगाँव रोड, सेक्टर-14, गुरुग्राम, हरियाणा -122001, भारत
हमारे पास उपलब्ध टेस्ट
- सीबीसी (कम्पलीट ब्लड काउंट)
- एफबीएस (फास्टिंग ब्लड शुगर)
- थायरॉइड प्रोफाइल टोटल (T3, T4 और TSH)
- HbA1c (ग्लायकोसिलेटेड हीमोग्लोबिन)
- पीपीबीएस (पोस्टप्रैन्डियल ब्लड शुगर)
- लिपिड प्रोफाइल
- विटामिन डी (25-हाइड्रॉक्सी)
- यूरिन आर/एम (यूरिन रूटीन और माइक्रोस्कोपी)
- कोरोनावायरस कोविड -19 टेस्ट- RT PCR
- एलएफटी (लिवर फंक्शन टेस्ट)
- केएफटी (किडनी फंक्शन टेस्ट)
- टीएसएच (थायरॉयड स्टिमुलेटिंग हार्मोन) अल्ट्रासेंसिटिव
- ईएसआर (एरिथ्रोसाइट सेडिमेंटेशन रेट)
- यूरिक एसिड, सीरम
- विटामिन B12
- सीआरपी (सी-रिएक्टिव प्रोटीन), क्वांटिटेटिव
- यूरिन सी/एस (यूरीन कल्चर और सेन्सिटिविटी)
- सीरम इलेक्ट्रोलाइट्स
- सीरम कैल्शियम
- सीरम क्रिएटिनिन
- डायबिटीज स्क्रीनिंग (HbA1C और फास्टिंग शुगर)
- इलेक्ट्रोलाइट्स के साथ केएफटी (इलेक्ट्रोलाइट्स के साथ किडनी फंक्शन टेस्ट)
- कोलेस्टेरॉल - टोटल
- एचबीए (हेमोग्लोबिन)
- कम्प्लीट हीमोग्राम (सीबीसी और ईएसआर)